In living organisms, gene expression is a complex process that results in the production of proteins from the genome in a time- and space-regulated manner. This expression is strictly dependent on the synthesis of messenger RNAs transcripts from the DNA molecule and their subsequent translation into proteins by the ribosomes. As they are synthesized, mRNAs undergo chemical modifications that ensure their stability, their delivery to ribosomes and eventually their translation into proteins. These modifications include the addition of a cap at the 5' end and polyadenosines tail at the 3' end, as well as the addition of methyl groups on various nucleotides. These numerous modifications provide an additional level of information for the control of protein production and collectively form the epi-transcriptome.
Researchers at IRIG, in collaboration with the University of Geneva, focused on a RNA mammalian methyltransferase, the PCIF1 protein (homologous to the Drosophila Pcif1 protein), which adds an extra methyl group to m6A (m6 adenosine) to form m6Am when the first transcribed nucleotide is an adenosine. In mice, mutation of PCIF1 encoding gene causes deregulation of the expression of a set of genes and a reduced body weight. During evolution, this protein naturally lost its catalytic activity in Drosophila where Pcif1 is, like its human counterpart, expressed in the nucleus and associated with the C-terminal domain of RNA polymerase (RNA Pol II). The work of the scientists shows that mutation of
Pcif1 gene in
Drosophila results in a deregulation of the expression of a set of genes, reduced body weight and a significant drop in fertility which indicates an important role for this protein in the physiology of the organism. In agreement with these observations, the Pcif1 protein is distributed all along the
polytene chromosomes at the level of active transcription sites and interferes with chromatin-dependent gene expression regulation. This protein binds to the phosphorylated form of serine 5 of RNA polymerase II (Figure) and may directly modulate its activity or promote the recruitment of chromatin components. These results suggest a similar contribution of PCIF1 to the fine regulation of RNA polymerase II activity in mammals, in addition to its mRNA methylation activity, whose role in mRNA translation efficiency is actively studied but still controversial.
This study demonstrates the importance to not restrict the function of enzymes to their sole catalytic role in the living world and the interest of thoroughly examining the evolution of proteins in eukaryotes in order to discover unsuspected or hidden mechanisms of action.
A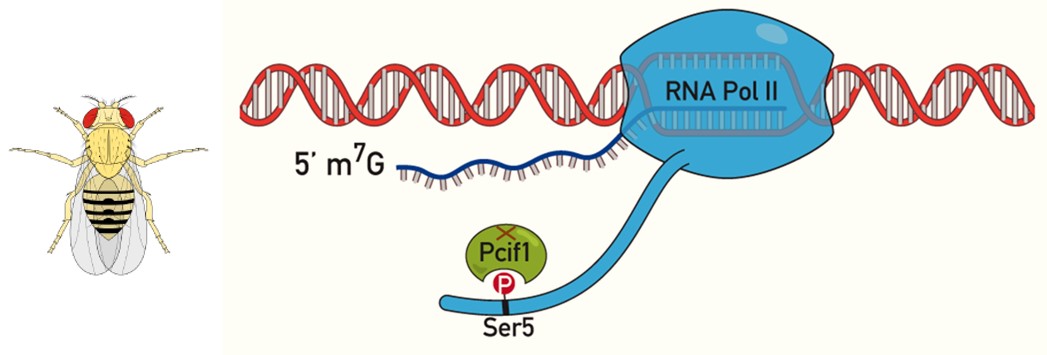
The lack of catalytic activity of Pcif1 in Drosophila reveals a new mechanism of gene expression regulation.
Figure A: The mammalian PCIF1 catalyses methylation (m6Am) of the first transcribed nucleotide of a messenger RNA, when it is an adenosine, however its fly
PCIF1 counterpart is naturally catalytically-dead. The fly Pcif1 expressed in the nucleus and binds the phosphorylated C-terminal domain (CTD) of RNA Pol II (at the level of phosphorylated serine 5).
Figure B: Immunofluorescence analysis with anti-Pcif1 antibodies revealed the presence of the Pcif1 protein (in green) along the chromosome at transcriptionally active euchromatin sites (i.e. excluded from DAPI dense staining, in red).
Based on work described in Pandey
et al. 2020; Franco
et al. 2023.
Polytene chromosomes result from a succession of chromatid duplications that remain associated without concomitant cell division. They are made up of several hundred or thousands of chromatids. Regions of condensed chromatin can be observed (called heterochromatin, in red on the figure), and less condensed regions of chromatin (called euchromatin) which correspond to sites of active transcription of DNA into RNA.
This work has been funded by the LabEX GRAL, ANR-10-LABX-49-01 and IDEX PhD international grant (to GF) financed within the University of Grenoble Alpes graduate school (Ecoles Universitaires de Recherche) CBH-EUR-GS (ANR-17-EURE-0003) and the Région Rhône-Alpes (international program).