The central mechanisms affecting the life of bacteria are controlled by complex regulatory networks allowing adaptation to a constantly changing environment. The bacterial genus Pseudomonas comprises more than a hundred species with exceptional adaptive capacities. Widespread in the environment, they are present in many different ecosystems, such as soil, water, plants, animals and even clouds. This adaptation generally involves reprogramming the expression of newly acquired genes in order to adjust their expression to the specific needs of the environment encountered by the recipient bacteria.
In previous studies, IRIG researchers have identified the Exolysin A protein as a major virulence factor in different
Pseudomonas species. This toxin causes cell death through the formation of pores in the membranes of its host. It is encoded by an
operon (operon
exlBA) acquired by
horizontal gene transfer. Because of the diversity of environments in which these bacteria live, these researchers, in collaboration with a team from Harvard Medical School (Boston, USA), have been interested in the regulation of the synthesis of this toxin in several species of
Pseudomonas.
By studying different clinical strains of
P. aeruginosa, they discovered the existence of ErfA, a transcriptional factor involved in the repression of the operon
exlBA. The study of the ErfA
regulon shows that the regulator's main target is unrelated to Exolysin A and toxicity. To get insights into this co-regulation of two different functions, they studied the ErfA regulon across several
Pseudomonas species and found that ErfA regulation of the
exlBA operon, and therefore virulence, is specific to the
P. aeruginosa species.
The researchers then studied the
regulatory regions of the
exlBA operon of 446 strains of
Pseudomonas (Figure). This analysis allowed them to show that certain regulatory elements appeared specifically in
P. aeruginosa, whereas other
Pseudomonas species have predicted patterns to be recognized by other regulators.
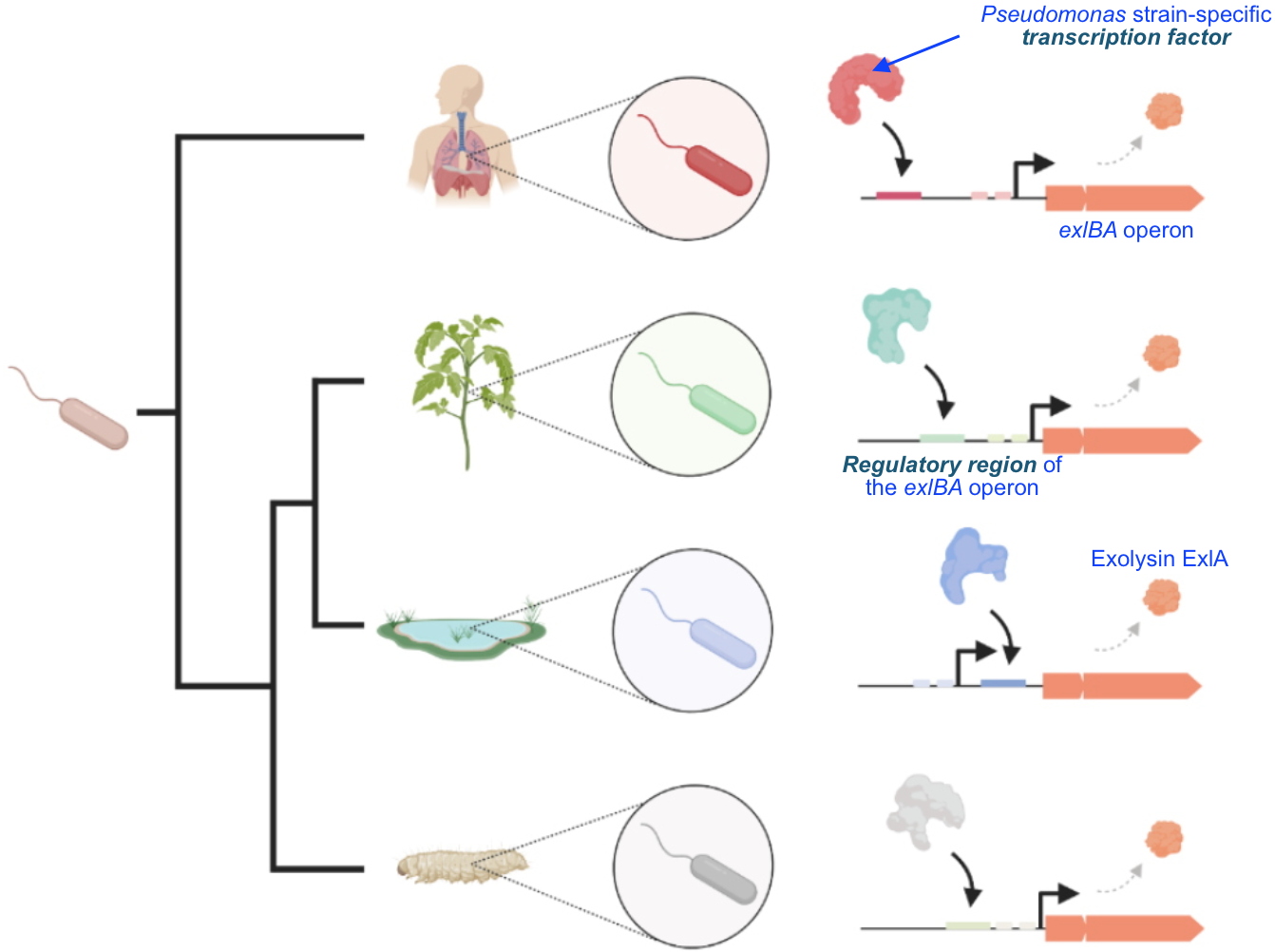
Illustration of the diversity of gene regulation in bacteria living in different environments.
Each strain has developed specific regulatory sequences for the
exlBA genes allowing the recruitment of different transcription factors that ensure an expression more adapted to specific environments (lung, plants, stagnant water, etc).
This work illustrates an adaptation mechanism of bacteria to different environments. After the acquisition of new genes, the evolutionary pressure would become strong on their regulatory sequences. This would force the selection of specific regulatory binding sites and consequently an adapted expression through pre-existing signaling pathways.
An
operon is a cluster of co-transcribed genes under the control of the same promoter.
Horizontal gene transfer is a process in which one organism integrates genetic material from another organism.
A
regulon is a group of genes whose expression is co-regulated by the same transcription factor.
A
transcription factor is a protein required to initiate or regulate the transcription of a gene.